- Super User
- Trials
- Read Time: 1 min
- Hits: 0
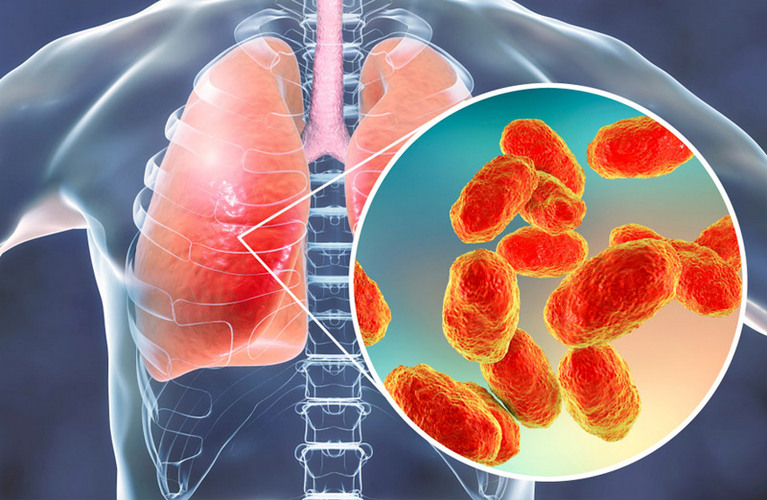
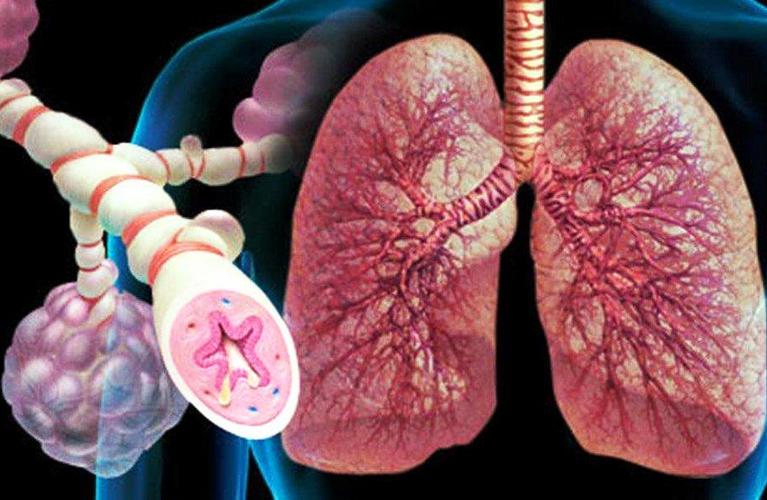
Recruitment for a clinical trial of a new Russian HIB vaccine for children from 3 to 6 months has begun. The comparison drug is Pentaxime, the placebo group is not assumed. All examination and supervision of a pediatrician is free of charge.